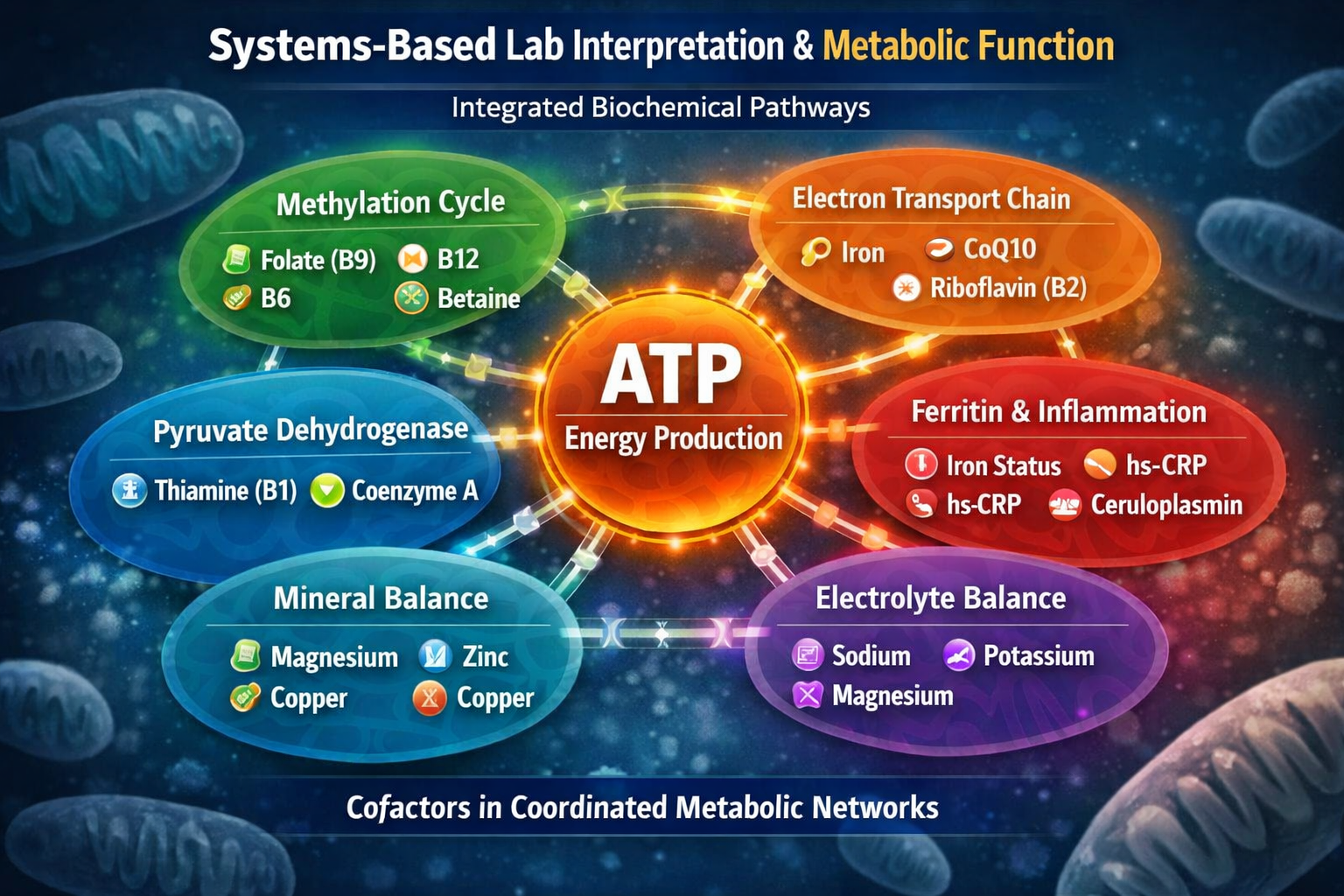
Why Nutrients Work in Clusters — Not Isolation
A Systems-Based Approach to Lab Interpretation and Metabolic Function
Most supplement strategies are reductionist.
Low iron? Take iron.
High homocysteine? Take folate.
Fatigue? Take B12.
But cellular metabolism does not operate on single nutrients. It operates through integrated biochemical pathways requiring multiple enzymatic cofactors functioning simultaneously.
Understanding nutrient clusters is essential for interpreting lab patterns, mitochondrial function, inflammation, and metabolic resilience.
Biology Is Pathway-Based, Not Ingredient-Based
Enzymes require cofactors.
Cofactors require transport systems.
Transport systems depend on redox state and mineral balance.
When one nutrient is marginal, a pathway may still function.
When several are marginal, metabolic efficiency declines — even if laboratory values remain within reference ranges.
This concept is foundational to the Metabolic Nutrient Framework, which connects laboratory markers to functional biochemical systems.
ATP Production Requires Multi-Nutrient Support
Mitochondrial ATP generation depends on coordinated activity across:
-
Pyruvate dehydrogenase (Thiamine-dependent)
-
Electron transport chain (Riboflavin, Niacin)
-
Coenzyme A synthesis (Pantothenic acid)
-
Iron-containing cytochromes
-
Magnesium-bound ATP (Mg-ATP complex)
Magnesium binds ATP to create biologically active Mg-ATP, the true substrate used in energy transfer reactions (de Baaij et al., 2015 – PMID: 21559048).
Iron is essential for cytochrome function within the electron transport chain and oxidative phosphorylation (Beard, 2001 – PMID: 11160540).
Thiamine deficiency impairs pyruvate dehydrogenase and mitochondrial respiration (Lonsdale, 2006 – PMID: 16651026).
ATP synthesis is therefore not dependent on one nutrient, but a coordinated network.
Homocysteine Is a Systems Marker
Homocysteine metabolism involves:
-
Folate (5-MTHF)
-
Vitamin B12
-
Vitamin B6
-
Riboflavin (B2)
-
Betaine (choline-derived)
Meta-analyses demonstrate that folic acid reduces homocysteine in a dose-dependent fashion, but riboflavin status significantly modifies response in individuals with MTHFR polymorphisms (Homocysteine Lowering Trialists’ Collaboration – PMID: 16567642).
Riboflavin acts as a cofactor for MTHFR. Without adequate B2, folate metabolism is impaired.
Elevated homocysteine is not simply a folate problem — it reflects integrated one-carbon metabolism.
For deeper pathway context, see:
👉 Educational Blood Lab Interpretation
👉 Metabolic Nutrient Framework
Ferritin Is Influenced by Inflammation
Ferritin is both an iron storage protein and an acute-phase reactant.
During inflammatory signaling, ferritin increases independent of iron sufficiency (Kell & Pretorius, 2014 – PMID: 24617844).
This means:
-
Ferritin can be elevated with inflammation
-
Functional iron delivery may still be limited
-
Oxygen transport and mitochondrial function may remain suboptimal
Iron metabolism also interacts with copper-dependent ceruloplasmin for iron mobilization (Hellman & Gitlin, 2002 – PMID: 12011125).
Thus, iron status is not isolated from inflammation, redox biology, or mineral balance.
Zinc and Copper Interact
High-dose zinc supplementation induces metallothionein, which preferentially binds copper and can reduce systemic copper status (Fosmire, 1990 – PMID: 2407097).
Copper is required for:
-
Cytochrome c oxidase (Complex IV of the ETC)
-
Iron mobilization
-
Superoxide dismutase (Cu/Zn SOD)
Excessive zinc without copper monitoring may impair mitochondrial function indirectly.
This illustrates how a “simple” nutrient intervention can alter multiple systems.
Electrolytes Function as a Regulatory Network
Hydration is not synonymous with water intake.
Sodium and potassium regulate membrane potential and cellular volume, while magnesium modulates ATP stabilization and neuromuscular excitability.
Low sodium levels correlate with fatigue, cognitive dysfunction, and orthostatic symptoms in certain populations (Verbalis et al., 2013 – PMID: 23649864).
Electrolytes operate as an integrated physiological system.
Why This Matters for Lab Interpretation
Routine lab panels measure downstream outputs:
-
Ferritin
-
Homocysteine
-
hs-CRP
-
Electrolytes
-
Lipid markers
But these reflect upstream pathway efficiency.
When multiple cofactors are borderline, laboratory values may remain technically “normal” while functional capacity declines.
A systems-based model evaluates:
-
Pathways
-
Cofactor dependencies
-
Inflammatory modifiers
-
Lab pattern clusters
This approach is structured in the:
👉 Metabolic Nutrient Framework
Key Takeaway
Nutrients do not function independently.
They operate within:
-
Mitochondrial enzyme complexes
-
One-carbon metabolism cycles
-
Redox networks
-
Mineral transport systems
-
Hormonal regulatory loops
Correcting a single nutrient without evaluating its supporting cofactors may produce incomplete results.
Biology is network-based.
Lab interpretation must be network-based as well.
FAQ (Snippet-Optimized)
Why do supplements sometimes stop working?
Because biochemical pathways require multiple cofactors. Addressing one nutrient without supporting the system may limit sustained benefit.
Why is homocysteine elevated despite folate supplementation?
Because homocysteine metabolism also depends on B12, B6, riboflavin, and methyl donors.
Can ferritin be elevated due to inflammation?
Yes. Ferritin acts as an acute-phase reactant and may rise independent of iron sufficiency.
Why is magnesium important for energy?
Magnesium binds ATP to form Mg-ATP, the biologically active form required for cellular energy transfer.
Stronger Authority CTA
If you want to understand how laboratory markers connect to mitochondrial energy, methylation, inflammation, and mineral regulation, explore the full systems-based model:
👉 Metabolic Nutrient Framework
That framework organizes nutrients by pathway, evidence tier, lab associations, and safety considerations — moving beyond isolated supplementation toward structured metabolic interpretation.
References
-
de Baaij JHF, Hoenderop JGJ, Bindels RJM. Magnesium in man: implications for health and disease. Physiol Rev. 2015;95(1):1–46.
https://doi.org/10.1152/physrev.00012.2014 -
Beard JL. Iron biology in immune function, muscle metabolism and neuronal functioning. J Nutr. 2001;131(2S-2):568S–579S.
https://doi.org/10.1093/jn/131.2.568S -
Lonsdale D. A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives. Evid Based Complement Alternat Med. 2006;3(1):49–59.
https://doi.org/10.1093/ecam/nek009 -
Homocysteine Lowering Trialists’ Collaboration. Dose-dependent effects of folic acid on blood homocysteine concentrations: a meta-analysis of randomized trials. Am J Clin Nutr. 2005;82(4):806–812.
https://doi.org/10.1093/ajcn/82.4.806 -
Kell DB, Pretorius E. Serum ferritin is an important inflammatory disease marker. Metallomics. 2014;6(4):748–773.
https://doi.org/10.1039/C3MT00347G -
Hellman NE, Gitlin JD. Ceruloplasmin metabolism and function. Annu Rev Nutr. 2002;22:439–458.
https://doi.org/10.1146/annurev.nutr.22.012502.114457 -
Fosmire GJ. Zinc toxicity. Am J Clin Nutr. 1990;51(2):225–227.
https://doi.org/10.1093/ajcn/51.2.225 -
Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med. 2013;126(10 Suppl 1):S1–S42.
https://doi.org/10.1016/j.amjmed.2013.07.006


